Scientist Wim L. Noorduin Coaxes Chemicals to Beautiful Blooms
Meghan Young — May 24, 2013 — Tech
References: visualnews
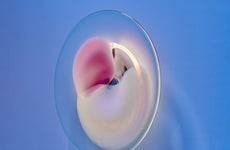
At first, a person might look at Wim L. Noorduin's images and assume that he is a talented macro photographer. In reality, he is a Harvard scientist who has learned how to manipulate microscopic chemicals to form beautiful blooms anyone would want to pluck on put on the dining room table. Adept at arranging crystals into leaves and petals, Wim L. Noorduin has created works of art that could be hung on the walls of a gallery.
Although Wim L. Noorduin makes the process seem easy, Harvard School of Engineering and Applied Sciences reports, "[He and] his colleagues dissolve barium chloride (a salt) and sodium silicate (also known as waterglass) into a beaker of water. Carbon dioxide from air naturally dissolves in the water, setting off a reaction which precipitates barium carbonate crystals. As a byproduct, it also lowers the pH of the solution immediately surrounding the crystals, which then triggers a reaction with the dissolved waterglass." Sounds pretty complicated.
Although Wim L. Noorduin makes the process seem easy, Harvard School of Engineering and Applied Sciences reports, "[He and] his colleagues dissolve barium chloride (a salt) and sodium silicate (also known as waterglass) into a beaker of water. Carbon dioxide from air naturally dissolves in the water, setting off a reaction which precipitates barium carbonate crystals. As a byproduct, it also lowers the pH of the solution immediately surrounding the crystals, which then triggers a reaction with the dissolved waterglass." Sounds pretty complicated.
Trend Themes
-
Microscopic Chemical Manipulation — The trend of manipulating microscopic chemicals to create beautiful blooms or artworks has the potential for disruptive innovation in the fields of art, interior design, and decorative accessories.
-
Crystal Arrangements — The trend of arranging crystals into leaves and petals opens up opportunities for disruptive innovation in the fields of jewelry design, fashion accessories, and home decor.
-
Chemical Reactions and Art — The trend of using chemical reactions to create art can lead to disruptive innovation in the fields of fine art, sculpture, and installation art.
Industry Implications
-
Art — The art industry can explore innovative ways of utilizing microscopic chemical manipulation to create unique and visually stunning artworks.
-
Interior Design — The interior design industry can incorporate crystal arrangements into their designs, offering a new and captivating element to spaces.
-
Science Education — The field of science education can utilize the concept of chemical reactions and art to engage students in creative and hands-on learning experiences.
3.1
Score
Popularity
Activity
Freshness