FDA Recalls Hydroxycut for Liver Damage
Marissa Brassfield — May 1, 2009 — World
References: cnn
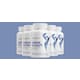
The Hydroxycut line of dietary supplements is being recalled by the FDA after they received 23 reports of serious liver injuries from Hydroxycut users, according to CNN. The FDA has instructed consumers to cease their use of the supplements immediately.
Hydroxycut has been linked to the death of a 19-year-old man, as well as multiple reports of liver damage, liver failure, cardiovascular issues, seizures and jaundice.
Implications - The chief medical officer of the FDA's Center for Food Safety and Applied Nutrition named Dr. Linda Katz says that when dealing with Hydroxycut, "adverse events are rare, but exist. Consumers should consult a physician or other health care professional if they experience symptoms possibly associated with these products."
Hydroxycut has been linked to the death of a 19-year-old man, as well as multiple reports of liver damage, liver failure, cardiovascular issues, seizures and jaundice.
Implications - The chief medical officer of the FDA's Center for Food Safety and Applied Nutrition named Dr. Linda Katz says that when dealing with Hydroxycut, "adverse events are rare, but exist. Consumers should consult a physician or other health care professional if they experience symptoms possibly associated with these products."
Trend Themes
-
Dietary Supplement Safety — With the FDA recall of Hydroxycut due to serious liver injuries, there will be a heightened focus on the safety of dietary supplements.
-
Natural Alternatives to Supplements — One potential disruptive innovation opportunity could be the development of natural alternatives to supplements with dangerous side effects.
-
Personalized Nutrition Plans — The danger of using dietary supplements underscores the importance of personalized nutrition plans to ensure individuals are receiving adequate nutrients and not resorting to potentially harmful supplements.
Industry Implications
-
Supplement Industry — The FDA recall of Hydroxycut raises questions about the safety and regulation of the supplement industry.
-
Healthcare Industry — With the potential for serious health risks associated with dietary supplements, there could be increased opportunities for healthcare professionals and organizations to provide guidance and support.
-
Organic and Natural Food Industry — The recall of supplements with harmful side effects may lead consumers to turn to organic and natural food options as a safer alternative for achieving their health goals.
1.1
Score
Popularity
Activity
Freshness