Withings Receives Approval for Its BeamO Device
References: insider.fitt.co
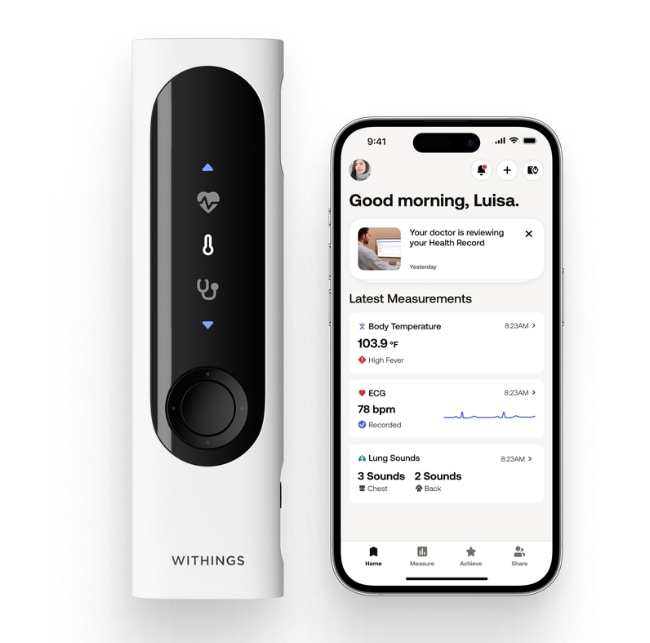
Withings is reportedly revolutionizing at-home family healthcare as its consumer health device — named BeamO — which integrates the functionalities of several discrete medical instruments into a single unit, recently received regulatory approval from the U.S. Food and Drug Administration.
BeamO is designed to perform several primary measurements — it conducts a single-lead electrocardiogram (ECG) via contact electrodes, captures heart and lung sounds through a digital stethoscope function, and measures body temperature using a non-contact infrared sensor. All recorded data is synchronized with a companion smartphone application, which includes features for securely sharing this information with healthcare providers and offers an optional subscription service that provides access to a professional review of ECG results by a cardiologist.
Image Credit: Withings
BeamO is designed to perform several primary measurements — it conducts a single-lead electrocardiogram (ECG) via contact electrodes, captures heart and lung sounds through a digital stethoscope function, and measures body temperature using a non-contact infrared sensor. All recorded data is synchronized with a companion smartphone application, which includes features for securely sharing this information with healthcare providers and offers an optional subscription service that provides access to a professional review of ECG results by a cardiologist.
Image Credit: Withings
6.3
Score
Popularity
Activity
Freshness