Peri Accurately Tracks Symptoms for Effective Management
Laura McQuarrie — April 8, 2026 — Tech
References: myperi.co & femtechinsider
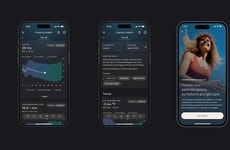
Peri, a first-of-its-kind perimenopause wearable that tracks symptoms and offers actionable insights, is now available for pre-order. Unlike wrist-worn devices or smart rings, this lightweight wearable adopts an under-breast position to accurately, discreetly and unobtrusively capture vasomotor symptoms.
There are no standardized biomarkers and no objective way to measure how perimenopause evolves, and Peri is building the first large-scale dataset on perimenopause. The continuous data collected by women in perimenopause is turned into useful personal information via a companion app, painting a picture of how sleep, mood, and activity levels all play a role in one's perimenopause journey—and Peri passively tracks hot flashes and night sweats so that users don't have to invest time and energy in manual logging.
There are no standardized biomarkers and no objective way to measure how perimenopause evolves, and Peri is building the first large-scale dataset on perimenopause. The continuous data collected by women in perimenopause is turned into useful personal information via a companion app, painting a picture of how sleep, mood, and activity levels all play a role in one's perimenopause journey—and Peri passively tracks hot flashes and night sweats so that users don't have to invest time and energy in manual logging.
Trend Themes
-
Under-breast Biometric Wearables — This form factor represents a novel approach to capturing localized vasomotor signals that enables higher-fidelity symptom detection than traditional wrist or ring devices.
-
Passive Symptom Tracking — Passive, continuous monitoring of hot flashes and night sweats presents an opportunity to move beyond subjective diaries toward objective, time-stamped symptom records.
-
Perimenopause Data Aggregation — Building Large-Scale Datasets On Perimenopause Creates New Foundations For Predictive Models That Correlate sleep, mood, and activity with symptom trajectories.
Industry Implications
-
Women's Health Technology — The convergence of form-factor innovation and symptom-specific sensing opens avenues for personalized care pathways and product ecosystems tailored to perimenopausal needs.
-
Digital Sleep and Wellness Apps — Integration of objective vasomotor data with sleep and activity metrics could reshape wellness offerings by enabling more precise symptom-context correlations in consumer apps.
-
Clinical Research and Pharmaceutical Development — Access To Continuous, Objective Symptom Data Offers A New Evidence Base That Can Inform Trial endpoints, drug-target validation, and stratified treatment approaches.
6.9
Score
Popularity
Activity
Freshness