Dermalogica Introduced the PRO Pen Microneedling System
References: prnewswire
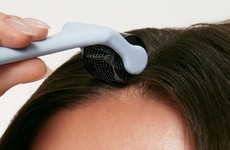
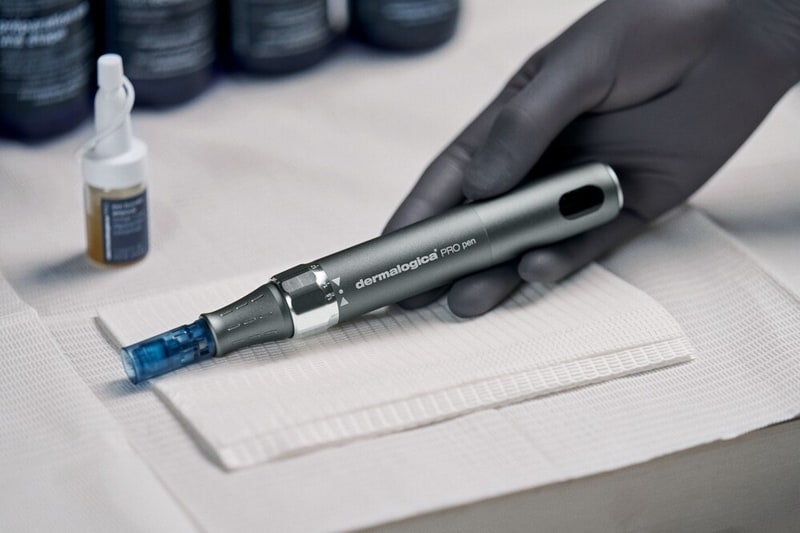
Dermalogica's latest PRO Pen Microneedling System represents the brand’s first FDA-cleared device and expands its professional portfolio into advanced in-office treatment technologies. This product innovation specifically aims to improve the appearance of facial acne scars in adults aged 22 and older.
Dermalogica's latest PRO Pen Microneedling System comes with accessories and has been manufactured to meet rigorous medical device standards. The device uses biocompatible materials and has also secured regulatory authorizations in Canada and Australia, with ISO 13485 certification obtained prior to approval in Canada. The PRO Pen integrates with Dermalogica’s existing ecosystem of professional treatments and at-home skincare, allowing licensed skin health professionals to offer microneedling procedures that can be paired with post-treatment products such as the Exo Booster system, which contains Lactobacillus-derived exosome-like vesicles designed to support skin regeneration and dermal remodeling.
Image Credit: Dermalogica
Dermalogica's latest PRO Pen Microneedling System comes with accessories and has been manufactured to meet rigorous medical device standards. The device uses biocompatible materials and has also secured regulatory authorizations in Canada and Australia, with ISO 13485 certification obtained prior to approval in Canada. The PRO Pen integrates with Dermalogica’s existing ecosystem of professional treatments and at-home skincare, allowing licensed skin health professionals to offer microneedling procedures that can be paired with post-treatment products such as the Exo Booster system, which contains Lactobacillus-derived exosome-like vesicles designed to support skin regeneration and dermal remodeling.
Image Credit: Dermalogica
Trend Themes
-
Fda-cleared Professional Devices — Certification Of Clinical-Grade Aesthetic Tools Signals Shifts Toward Medical-Level Innovation In Salon And Spa Services.
-
Biocompatible Consumables — The Use Of Medical-Grade Materials And Single-Use Accessories Points To New Supply Chains For Safer, Scalable In-Office Treatments.
-
Integrated Clinic-to-home Ecosystems — Connecting In-Office Procedures With Complementary Post-Treatment Products And At-Home Regimens Opens Pathways For Seamless Patient Journeys And Recurring Revenue Models.
Industry Implications
-
Medical Aesthetics — Clinics And Professional Brands Are Positioned To Expand Service Portfolios With Regulated Device Offerings Targeting Scarring And Skin Remodeling.
-
Dermal Biomaterials — Manufacturers Of Biocompatible Components And Exosome-Like Biologics Could Advance Formulations That Enhance Regeneration And Integration With Device Therapies.
-
Regulatory and Certification Services — Third-Party Testing, ISO Consulting, And Cross-Border Approval Expertise Become Critical As Aesthetic Devices Pursue Multinational Market Entry.
8.1
Score
Popularity
Activity
Freshness