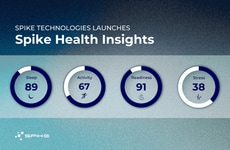
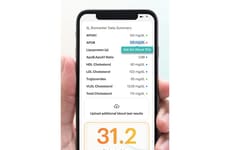
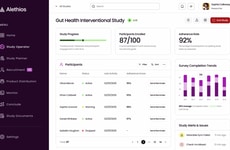
SAS Introduced a New Software Platform, SAS Clinical Acceleration
The SAS Clinical Acceleration platform is designed to address operational challenges within the clinical trial process for pharmaceutical and biotechnology companies. The system is constructed upon the SAS Viya data and AI platform. It functions as a centralized, cloud-native environment for managing, analyzing, and reporting clinical research information.
The SAS Clinical Acceleration platform's primary objective is to streamline the journey from initial data gathering to final regulatory submission for new therapies. A key feature of the solution is its focus on maintaining a validated and compliant workspace, incorporating audit trails, data traceability, and support for established regulatory data standards like CDISC, which are intended to mitigate the risk of noncompliance and associated delays.
The platform is engineered to be modular and supports integration with various existing systems, including electronic data capture tools and open-source technologies.
Image Credit: SAS
The SAS Clinical Acceleration platform's primary objective is to streamline the journey from initial data gathering to final regulatory submission for new therapies. A key feature of the solution is its focus on maintaining a validated and compliant workspace, incorporating audit trails, data traceability, and support for established regulatory data standards like CDISC, which are intended to mitigate the risk of noncompliance and associated delays.
The platform is engineered to be modular and supports integration with various existing systems, including electronic data capture tools and open-source technologies.
Image Credit: SAS
Trend Themes
-
Cloud-native Clinical Platforms — The rise of cloud-native platforms in clinical trials presents opportunities to enhance data management and analysis efficiency within pharmaceutical research.
-
Regulatory Compliance Automation — Automation of regulatory compliance processes using integrated systems can significantly reduce the risk of delays in drug approval times.
-
Modular Clinical Solutions — Modular systems that easily integrate with existing clinical tools facilitate a more adaptable and scalable approach for research organizations.
Industry Implications
-
Biotechnology — Biotechnology firms can leverage advanced data analytics solutions to streamline their complex clinical trial processes.
-
Pharmaceuticals — Innovative platforms that ensure compliance and data traceability offer pharmaceutical companies an edge in speeding up the drug development cycle.
-
Healthcare Software — The development of healthcare software solutions focused on clinical trials offers significant improvements in operational efficiencies and compliance management.
4.7
Score
Popularity
Activity
Freshness